
Intas Pharmaceuticals: Intas eyes Teva's oncology, women's health divisions in Europe - The Economic Times

Frontiers | Pooled Analysis on the Effectiveness and Safety of Lipegfilgrastim in Patients With Urological Malignancies in the Real-World Setting | Oncology

Lonquex - Lipegfilgrastim 10mg/ml - 1 Seringa Segurança Preenchida 0,6ml SC (2 A 8C) - Teva - ÁgilMed - Medicamentos Especiais e Nutrição Clínica

Immunogenicity assessment of PEGylated proteins, Lonquex, a PEGylated G-CSF case study - ScienceDirect
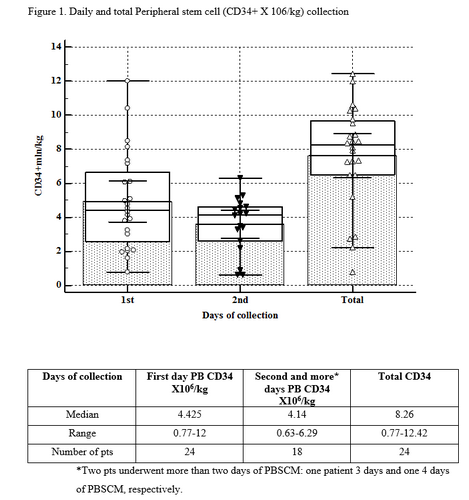
LONG-ACTING G-CSF (LIPEGFILGRASTIM, “LONQUEX”) FOR STEM CELL.... EHA Library. Danylesko I. Jun 15 2019; 267164

Immunogenicity assessment of PEGylated proteins, Lonquex, a PEGylated G-CSF case study - ScienceDirect


![Buy Lonquex® [Lipegfilgrastim] Buy Lonquex® [Lipegfilgrastim]](https://pillbuys.com/27724-home_default/toremifen.jpg)



![PDF) Role of lipegfilgrastim in the management of chemotherapy-induced neutropenia [Corrigendum] PDF) Role of lipegfilgrastim in the management of chemotherapy-induced neutropenia [Corrigendum]](https://i1.rgstatic.net/publication/277615466_Role_of_lipegfilgrastim_in_the_management_of_chemotherapy-induced_neutropenia_Corrigendum/links/55f6fe2b08aeba1d9eee5596/largepreview.png)




![Buy Lonquex® [Lipegfilgrastim] Buy Lonquex® [Lipegfilgrastim]](https://pillbuys.com/25251-large_default/lonquex-lipegfilgrastim-.jpg)




![Buy Lonquex® [Lipegfilgrastim] Buy Lonquex® [Lipegfilgrastim]](https://pillbuys.com/26185-home_default/oncotron-mitoxantrone-.jpg)