
Remoxy Advisory Committee Briefing Materials Form - Fill and Sign Printable Template Online | US Legal Forms

FDA Panels on Twitter: "Key excerpts of #FDA's briefing document for #sintilimab suggest broader regulatory applicability. FDA briefing document https://t.co/3dabfjaP1O Draft voting question https://t.co/6qW8xpiP3U All materials, including link to ...

Fillable Online fda Advisory committee briefing document - Food and Drug Administration - fda Fax Email Print - pdfFiller
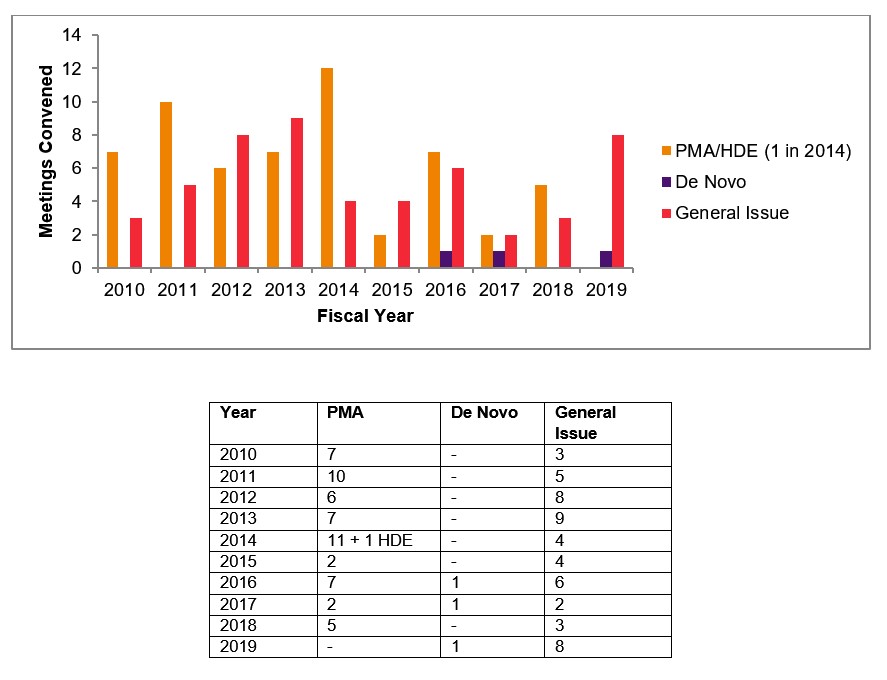
Bad News for Device Sponsors: Panel Meetings were Already Going the Way of the Homework Assignment, and COVID Might “Put the Nail in the Coffin” - Food and Drug Law Institute (FDLI)

Meg Tirrell on Twitter: "Proposed timing for Pfizer booster shot in FDA briefing documents is 6 months out from primary series - not 8, which was what US health officials suggested https://t.co/U2tCyCOfdQ" /

FDA advisory committee rejects recommending approval of lower-dose empagliflozin for type 1 diabetes

FDA advisory committee recommends not approving FibroGen's anemia drug candidate roxadustat - Drug Discovery and Development











